About PCORnet®
PCORnet is a national resource that offers the kind of research ecosystem that has long been pursued: a fully integrated network where vast, highly representative health data, research expertise, and patient insights are built-in and accessible from the very start. The infrastructure of PCORnet is well established, meaning that PCORnet® Network Partners know how to maximize the value of these connections to deliver fast, trustworthy answers that advance health outcomes.
Research
PCORnet is uniquely networked to offer the real-world evidence essential for answering the clinical questions that impact peoples' lives and support studies addressing major health conditions that affect a large number of Americans.
Data
With access to data from everyday healthcare encounters with more than 47 million people annually across the U.S., users of PCORnet have potential insights at their fingertips. These data are accessible via a distributed research network model of eight large PCORnet® Clinical Research Networks (CRNs) and facilitated by the Coordinating Center for PCORnet®.
PCORnet: An Introduction
Get an overview of PCORnet in this fact sheet and overview deck.
Learn about PCORnet milestones over the past decade in this timeline.
PCORnet® Dashboard
Review the PCORnet® Quarterly Dashboard
Q1-2026
Knock on the PCORnet® Front Door to access PCORnet
PCORnet is well suited to support the conduct of:
- Real-world evidence studies
- Comparative clinical effectiveness research
- Population health research
- Pragmatic research
- Health systems research
- And more
PCORnet®: Accelerating Patient-Centered Comparative Clinical Effectiveness Research
 A special February 2026 PCORI-sponsored supplement of the journal Medical Care looks at the scope and scale of PCORnet.
A special February 2026 PCORI-sponsored supplement of the journal Medical Care looks at the scope and scale of PCORnet.
Engagement
When patients and caregivers take on an active role in research, outcomes improve. That’s why every effort powered by PCORnet® broadly embeds the voice of patients and those who care for them. Patients and caregivers serve on the PCORnet® Steering Committee and provide leadership across every stage of research.
Network
PCORnet® CRNs not only source data, but also propel patient-centered research forward. PCORnet® CRNs include the nation’s leading clinical researchers whose collective knowledge and experiences empower and support users of the network.

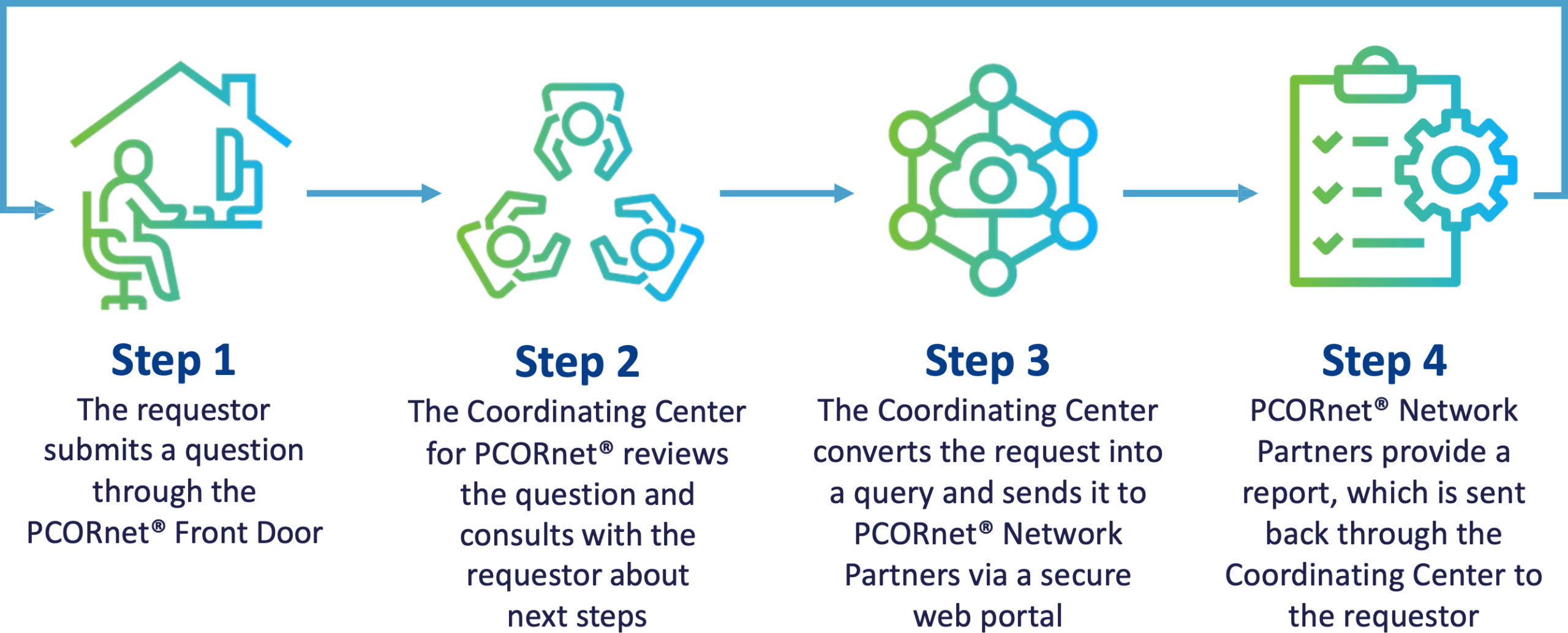
How It All Works
The PCORnet® Front Door is a central gateway point for anyone seeking to use PCORnet resources. Once users have reached out, the Coordinating Center for PCORnet® sets up a consultation to clarify the request and convert the question into a query to send out to the PCORnet® Network Partners for the best fit.